Carbon Capture and Sequestration (CCS)
Last modified: 12th August 2019
Is ‘carbon capture’ from coal possible?
When coal is burned, large amounts of carbon dioxide (CO2) are released into the atmosphere. Releases of CO2 from coal-fired power plants make up around 40% of total CO2 emissions in the US, and are therefore a major contributor to global climate change and ocean acidification. The aim of carbon capture and sequestration (CCS) is to reduce the climate change impact of burning coal and other fossil fuels. Theoretically, CCS could allow us to burn much of our remaining fossil fuel stores without continuing to release large quantities of CO2 into the air. In particular, this has been proposed as a “bridge” technology to be used to limit pollution while alternative sources of energy are developed. However, CCS is not expected to be ready to sequester a significant amount of carbon emissions until at least 2030 (IPCC 2005, DOE 20075.5 Mb) and most experts agree that serious mitigation of emissions will be needed before then.
The infrastructure required is expected to be very expensive and there are currently no coal-fired power plants in existence that use CCS, although small-scale pilot projects are being undertaken (see Further Reading). The only commercial-scale coal facilty in the US that uses CCS is a coal gasification plant in Wyoming that pumps the carbon dioxide to Canada for use in oil recovery. Permitting is also underway for a large-scale project called the Texas Clean Energy Project which hopes to capture and store 90% of the CO2 generated.
‘Clean Coal’?
The marketing term “clean coal” originated in reference to coal-fired power plants that implemented scrubbers to reduce emissions of the sulfur dioxides and nitrous oxides that contributed to acid rain. More recently, many people have used the term to refer instead to the idea of coal that utlizes CCS. The capture and sequestration technology discussed here would only address the problem of CO2 emissions and does not by itself affect the emissions of mercury, sulfur dioxide, and nitrous oxides.
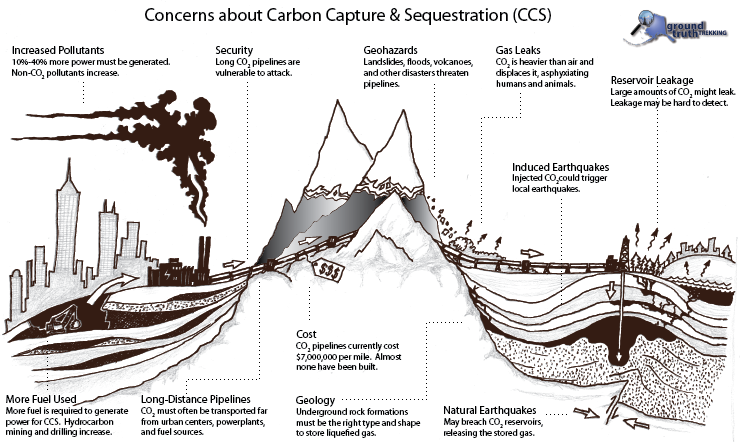
Problems Facing CCS
CCS consists of three seperate pieces, each of which has difficulties that must be overcome in order for the technology to become widespread, cost-effective, and safe.
Capture refers to removing carbon dioxide from other combustion gases and getting it into a storable form. Ninety nine percent of existing coal power plants in the US burn crushed “pulverized coal” that produces a variety of gaseous emissions. CO2 makes up only 10-15% of these emissions. In order to separate CO2 from the other gases, the emissions would need to be run through a filtration system (i.e. a “stack scrubber”), which would be difficult and expensive. There are alternative methods to burning pulverized coal, which would make CO2 separation easier, such as underground coal gasification and a pilot project using “oxyfuel”, but none of these technologies have been deployed on a large scale. It takes power to run stack scrubbers and compressors to pump CO2 to a storage site, meaning that if this technology were to be employed, 10-40% more coal would need to be burned to generate the same end use amount of electricity for consumers. Furthermore, scrubbing would not be completely efficient, meaning about 10-30% of the CO2 would still be emitted. Also, the increased coal consumed would lead to an increased production of other pollutants. Lastly, carbon capture would increase even further the large amounts of water required for coal combustion.
Transport would likely be in liquid form by pipeline from where the coal is burned to where the CO2 would be stored. In the US, we currently transport about 50 million metric tons of CO2 by pipeline annually (IPCC 2005, Chapter 4). This CO2 is produced in pure form by some industrial processes (so stack scrubbing is not needed), and is injected into oil and gas wells to enhance recovery. However, this existing capacity would need to increase to 3,600 million tons in order to move all the CO2 produced in the US. Pipelines to carry CO2 are very expensive, estimated to cost around $7 million for every 10 miles(Duke University, 20071 Mb; CRS, 2008). For example, to store just North Carolina’s CO2 would require a $500 million pipeline to a site with 3 years worth of storage, or a multi-state $5 billion pipeline project to a place with more storage (Duke University, 20071 Mb). Both transport and storage of CO2 on a large scale represent a potential health and environmental risk. CO2 suffocates animal life in high concentrations, and natural releases of CO23.7 Mb in the past have resulted in the death of forests, livestock, and humans.
Storage is currently being undertaken at a handful of natural gas and oil fields, where CO2 is injected into wells to enhance recovery of the oil and gas. If CO2 storage was to be undertaken on a large scale, sites would need to be chosen based on estimated storage capacity, proximity to the site of CO2 production, and geological factors, such as the presence of an impermeable surface layer and the ability of surrounding minerals to absorb CO2. Areas deep underground where saltwater fills rock pores, called “saline aquifers”, may present more storage opportunity. A difficult but safe option is to react CO2 with mafic rocks that have high amounts of iron and magnezium oxides to produce stable carbonate rocks like limestone. Lastly, CO2 could potentially be pumped into deep ocean trenches where it would pool and remain as a liquid. However, any leaks from sub-ocean sequestration would potentially acidify ocean waters and damage global marine ecosystems. In addition to the current logistics of storage, the CO2 would need to be stored “in perpetuity”.
Existing Projects
One of the world’s largest CO2 sequestration project is located in Norway. A large natural gas plant has pumped one million tons of CO2/year since 1996 below the ocean floor of the North Sea, into a reservoir that is capped with impermeable rock that keeps the CO2 from escaping. At this rate of sequestration, it would take 5-10 of these projects to store the CO2 emissions of a single large coal-fired power plant, and 3,600 to store CO2 emissions from all US emission sources. Other large projects include the Dakota coal gasification plant that transports around 3 million tons of CO2 per year to Canada for enchanced oil recovery and the In Salah gas project in Algeria which stores around 1 million tons of CO2 in a depleted gas resevoir.
No large-scale CCS projects are currently in operation using coal as a feedstock, although a number have been proposed. [CO2]: carbon dioxide [ CO2]: Carbon dioxide
Further Reading
- > Fundamentals of carbon capture and storage technology. 2008. The Petroleum Economist. (only available for purchase)
- > Massachusetts Institute of Technology. 2007. The future of coal: options for a carbon-constrained world.
- > US Department of Energy. Carbon Sequestration Technology Roadmap and Program Plan 2007.
- > Intergovernmental Panel on Climate Change. 2005. IPCC Special Report on Carbon Dioxide Capture and Storage.
- > A September 2009 Clean Air Task Force report on CCS entitled "Coal Without Carbon: An Investment Plan for Federal Action.
- > World Coal Association pages on CCS
- > "Carbon Capture and Sequestration Technologies" website by MIT
Created: Jan. 19, 2018